|
KOH + HNO3 -> KNO3 + H2O What is a Strong Base and a Weak Base?Ī strong base is a base which dissociates completely in water to yield hydroxide ion (OH-). They conduct electricity when dissolved in water.īases react with acids to form salts (Neutralization reaction) What are the properties of a base? Bases are slippery to touch and bitter to taste. 
Examples are H2SO3 (Sulphurous acid), HF (hydrofluoric acid), HNO2 (Nitrous acid) and C2H2O4 (Oxalic acid).īases are chemical compounds which dissociate in an aqueous solution to yield hydroxide ions (OH-). Weak acids are acids that ionize partially in an aqueous solution. Examples of strong acids are HCl (Hydrochloric acid), HNO3 (Nitric acid) and H2SO4 (Sulphuric acid). Strong acids are acids that ionize completely in an aqueous solution. Na2CO3 + H2SO4 -> Na2SO4 + H2O + CO2 What is a Strong Acid and Weak Acid? They react with metal carbonates to liberate carbon dioxide (CO2) gas. They have the ability to conduct electricity in an aqueous solution.Īcids react with metals to liberate hydrogen (H) gas. What makes acids peculiar? Acids have a sour taste. This means that they donate protons or hydrogen ions to other compounds. Since the compound is formed by a weak acid and a strong base, it is a basic salt.Ī substance which dissociates in an aqueous solution to yield hydrogen ions (H+) is known as an Acid. Potassium fluoride is a product of the neutralization reaction between hydrogen fluoride (HF) and potassium hydroxide (KOH). 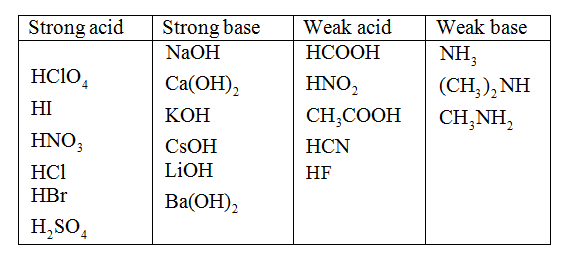
Now, you may be wondering – is potassium fluoride and acid or base? 
It can cause irritation and burns to the respiratory tract if inhaled. Potassium fluoride is hygroscopic and can be prepared by the neutralization of hydrogen fluoride (HF) by potassium hydroxide (KOH). It is commonly used as a fluoridating agent. It is soluble in water but insoluble in alcohol. Potassium fluoride (KF) is an alkali halide which appears as a white crystalline or powdery compound with a sharp saline taste and no odor.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
